Message

Masahiko Miyata
Chairman
Medical Technology Association of Japan
On the occasion of my appointment as Chairman of the Medical Technology Association of Japan (MTJAPAN), I would like to extend my sincere greetings.
The global struggle against COVID-19 has finally reached a turning point, and daily life is gradually returning to normal in both society and the healthcare field. We extend our deepest respect and appreciation to all medical professionals who worked tirelessly on the front lines, as well as to our member companies, who upheld the unwavering commitment to ensure uninterrupted access to essential medical devices.
The pandemic has accelerated the adoption of digital technologies and artificial intelligence (AI) across many sectors, including healthcare. With a projected shortage of medical professionals, improving the efficiency and quality of healthcare delivery has become a pressing issue. In response, the medical device industry is committed to developing innovative solutions that not only enhance clinical outcomes but also strengthen the broader healthcare delivery system. We will continue to pursue this through the introduction of advanced technologies and cross-sector collaboration.
While Japan's medical device exports are expanding, the industry also faces rising global challenges, including protectionist trade policies, tariff barriers, infectious disease risks, and geopolitical disruptions to supply chains. To remain competitive on the global stage, it is essential that we first establish a stable and sustainable business foundation domestically. To this end, our association will continue to work closely with government agencies to promote regulatory harmonization and supply chain resilience, thereby advancing the global development of the medical device industry.
From the standpoint of healthcare security, the expectations and responsibilities placed on the medical device sector have grown considerably. It is more critical than ever to build systems that ensure uninterrupted access to essential medical technologies, even in times of crisis. As an industry, we are committed to ongoing improvements and to strengthening our supply networks.
Regardless of external circumstances, upholding strict compliance and high ethical standards remains a fundamental responsibility. We recognize that earning the trust of society—especially that of patients and healthcare professionals—requires transparency, integrity, and a commitment to legal and ethical conduct across the entire industry.
Japan’s healthcare system faces increasing financial pressure due to an aging population. Maintaining high-quality medical care while optimizing costs is a national imperative. Leveraging Japan’s world-renowned strengths in technology and manufacturing quality, the medical device industry must take the lead in driving innovation and enhancing international competitiveness. By aligning with the strategic goals outlined in the “Vision for the Medical Device Industry” – developed by the Ministry of Economy, Trade and Industry and the Ministry of Health, Labour and Welfare – we will contribute to the creation of a sustainable healthcare system.
The Medical Technology Association of Japan remains committed to neutrality as an industrial body, while contributing to the advancement of the medical device sector and the healthcare system as a whole. We sincerely ask for the continued understanding and cooperation of our member companies, healthcare professionals, government agencies, and all related stakeholders.
Overview
Promptly providing safe and innovative medical technology Contributing to improved medical quality and the development of the medical device technology industry
Summary
Medical Technology Association of Japan
【Regular members】 267 companies
【Associate members】 42 companies
【Number of employees】
Japan:Approx. 79,500
Outside Japan: Approx. 87,500
As of June 2025
History
- 1967:
- Medical Plastics Conference established. 1990:Name changed to Japan Association of Medical and Material Industries (JAMMI)
- 1979:
- Japan Artificial Organ Industry Association (JAOIA) established
- 2000:
- JAMMI and JAOIA merged. Japan Medical Devices Manufactures Association (JMED) launched
- 2013:
- Reorganized as Medical Technology Association of Japan (MTJAPAN)
- 2023:
- 10th anniversary of Medical Technology Association of Japan (MTJAPAN) Establishment
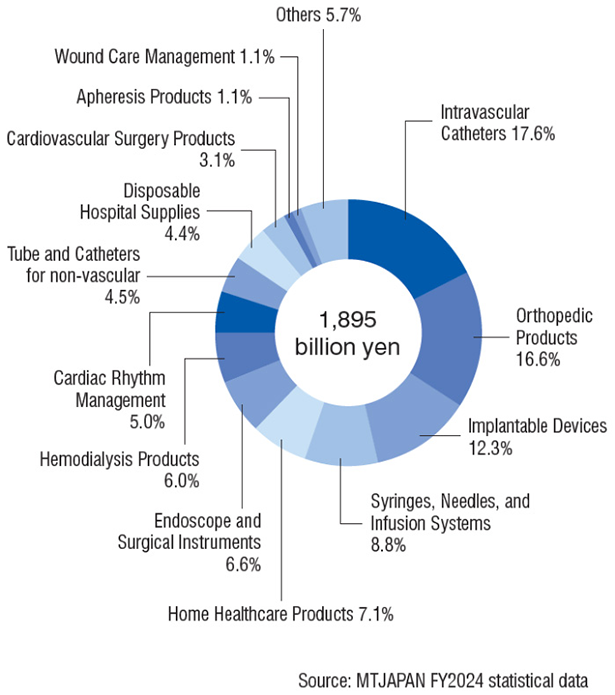
Composition of Domestic Shipment for MTJAPAN products
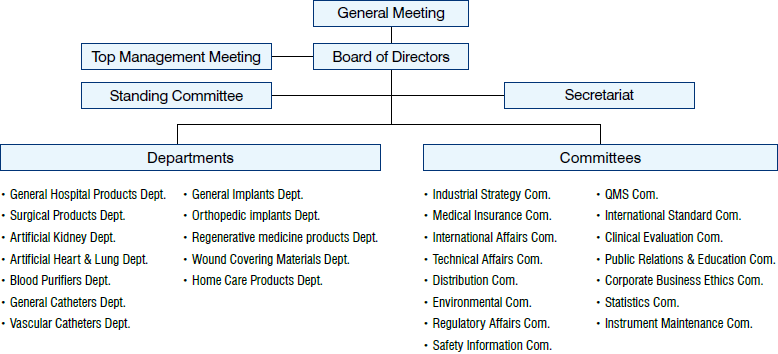
Organization
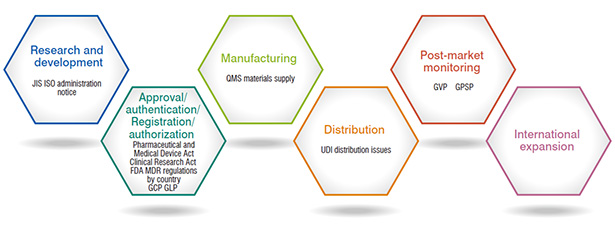
Activity
Development of business from R&D to post-commercialization and international expansion
- To maintain the effectiveness and safety of medical devices
- Maintaining stable supply
Business for member companies
- New administration and industry trends declarations
- Hosting of training seminars and lectures, etc., for members only
- Presenting opportunities to configure networks with the industry and government, etc.
- Proposing corporate requirements to government as a voice for the industry
- Other businesses for members
Publishing MTJAPAN news, and compiling MTJAPAN medical device statistical data
Operating medical technology seminars
Introducing MTJAPAN group insurance (product liability insurance both in Japan and other countries)
Assuring stable supplies during a disaster
Announcing government and industry trends
We periodically distribute notices from the government and offices on the MTJAPAN members website.
Website top page
Lectures and seminars
We organize lectures and seminars on the latest regulatory trends and business and employee training for companies.
- Lecture on medical insurance system
- Lecture on medical device regulations
- Lecture on clinical evaluation system
- Lecture on QMS compliance survey
- Lecture on trends in international standards
- Medical device technology seminar
- Overseas business expansion seminar
- Medical device distribution seminar
- Environmental regulation seminar
- Public relations seminar
- Post market surveillance workshop
- Corporate ethics education workshop
- Continuous training for selling and repairing medical devices*
(*Held in the capacity of a Ministry of Health, Labour and Welfare registered organization)

Post market surveillance workshop

Medical device technology seminar

General meeting

New Year's gatherings
Domestic and foreign networking
We provide networking opportunities with the medical device industry and government through employee general meetings/gatherings, New Year's gatherings, committees, and group activities.
We create global networks. Our activities include sending representatives to international conferences like AdvaMed, APACMED and GHWP, as well as hosting medical device symposiums.
Announcing policy proposals
Through committee activities, we announce policy proposals following discussions and meetings with the Japan Federation of Medical Devices Associations (JFMDA) and the government regarding policies concerning industrial development, health insurance systems, laws and regulations, and standards.

Gathering details
Participation in setting ISO and JIS standards
As the Japanese consultative body for the technical committee of four ISO fields, we send representatives to international conferences and participate in setting international standards.
- ISO/TC76 (Blood transfusion and infusion equipment, etc.)
- ISO/TC84 (Injection equipment, etc.)
- ISO/TC150 (Artificial lungs, surgical implant devices, etc.)
- ISO/TC194 (Biological and clinical evaluation of medical devices)
We have participated in establishing approx. 90 JIS standards concerning our organization's products.

ISO/TC194 International Conference
Publishing printed materials
We edit and publish materials concerning medical devices. We also introduce activities statuses and data through our website.
- Special Insurance Medical Materials Guidebook
Posting use objectives, methods, fees, and redemption prices, etc., for special insurance medical materials - MTJAPAN medical devices statistical data
Posting market and industry trends within the scope of our organization - MTJAPAN news (website)
Introducing activities statuses for our organization - Medical device past and present stories (website)
Introducing the history of various medical devices

Special Insurance Medical Materials Guidebook

MTJAPAN medical devices
statistical data
